Lower Baseline Germinal Center Activity and Preserved Th1 Immunity are
Associated with Hepatitis B Vaccine Response in Treated HIV Infection
Published March 14, 2017
Read in PDF Format
Authors
Robert M. Paris1,2, Lucimar G. Milagres2,3, Eirini Moysi4, Jason F. Okulicz5, Brian K. Agan6,7, Anu Ganesan6,7, Constantinos Petrovas4*, Richard A. Koup2*
Affiliated Institutions
1 US Military Malaria Research Program, Walter Reed Army Institute of Research, Silver Spring, Maryland2 Immunology Laboratory, Vaccine Research Center, NIAID, NIH, Bethesda, Maryland
3 Department of Microbiology and Immunology, Universidade do Estado do Rio de Janeiro, Rio de Janeiro, Brazil
4 Tissue Analysis Core, Immunology Laboratory, Vaccine Research Center, NIAID, NIH, Bethesda, Maryland
5 Infectious Diseases Service and HIV Medical Evaluation Unit, San Antonio Military Medical Center, San Antonio, Texas
6 Infectious Diseases Clinical Research Program, Department of Preventive Medicine and Biostatistics, Uniformed Services University of the Health Sciences, Bethesda, Maryland
7 Henry M. Jackson Foundation for the Advancement of Military Medicine, Bethesda, Maryland
*Equal contribution
Corresponding Author
Constantinos PetrovasVaccine Research Center, NIAID, NIH
Building 40
40 Convent Drive
Bethesda, MD 20892-3005
301-594-8573
petrovasc@mail.nih.gov
DOI
10.20411/pai.v2i1.175Suggested Citation
Paris RM, Milagres LG, Moysi E, Okulicz JF, Agan BK, Ganesan A, Petrovas C, Koup RA. Lower Baseline Germinal Center Activity and Preserved Th1 Immunity are Associated with Hepatitis B Vaccine Response in Treated HIV Infection. Pathogens and Immunity. 2017;2(1):66-88. doi: 10.20411/pai.v2i1.175Abstract
Background: Why HIV-infected individuals have poor responses to standard dose and schedule hepatitis B virus immunization is not well understood.
Methods: We compared the serologic and cellular immune profiles of treated HIV-infected individuals with similar durations of infection and preserved CD4 counts (> 350 cells/microliter) by hepatitis B vaccine (HBV) response before and after vaccination.
Results: Similar levels of immune activation and plasma cytokine profile were found between non-responders and responders. The baseline plasma levels of CXCL-13, a surrogate of germinal center reactivity, were significantly lower in HBV responders compared to HBV non-responders and were a predictor of both vaccine response and titer. Furthermore, response to HBV vaccination was associated with a significantly higher frequency of circulating IgGhigh memory B cells post vaccination and preserved Th1 antigen-specific T-cell responses.
Conclusions: Taken together, our data suggest that preserved Th1 responses are associated with hepatitis B vaccine response in treated HIV infection.
Keywords: Human immunodeficiency virus, AIDS, hepatitis B vaccine, T cell, antibodies
Introduction
Approximately 5-10% of healthy adults have an inadequate response to three doses of hepatitis B vaccine [1], which is associated with human leukocyte antigen (HLA) class II polymorphism [2, 3]. Hepatitis B vaccine (HBV) non-response (defined as hepatitis B surface antibody < 10 mIU/mL after completion of the three-immunization series) is even more common in HIV infection [4, 5], and immune reconstitution with combination antiretroviral therapy (cART) appears to have less impact on this response [4, 6]. This is in contrast to influenza vaccination where vaccine response and protection are associated with CD4 count and cART [7-10]. Even with a four-vaccination, double-dose immunization regimen, a significant number of HIV-infected individuals failed to demonstrate an adequate immune response [11]. Importantly, the timing of vaccination relative to HIV infection (before vs after) appears to impact protection from hepatitis B infection: Landrum et al. documented significantly more hepatitis B breakthrough infections among HIV-infected individuals who were vaccinated after HIV infection and despite an HBV titer greater than 10 mIU/ml [4]. These findings suggest that the immune response to HBV among HIV-infected individuals may be qualitatively different than the immune responses to influenza or other subunit vaccines. The inability to mount or maintain memory B-cell responses is a well-recognized phenomenon in chronic HIV infection, and a number of studies have demonstrated the loss of serological memory to immunization [12] and CD27high memory B cells [13, 14], which most likely begins at the time of infection and continues throughout the course of the disease (reviewed in [15]). The concurrent loss of both T-dependent and T-independent antigen-specific responses suggests a direct effect of HIV on B-cell populations and both direct and indirect effects on T-B cell interactions [14]. Potential mechanisms for B-cell dysfunction include direct viral effects on B-cells as well as polyclonal activation, resulting in B-cell exhaustion and altered homing [16], and impaired class switch recombination and affinity maturation [17]. In contrast, T-cell phenotype and function in HBV vaccination is not well studied. In the last decade, seminal observations of follicular T-B cell interactions have revealed a unique set of T cells that provides direct B-cell help [18] within secondary lymphoid tissues, referred to as T follicular helper cells (Tfh). These cells are typically characterized by expression of CXCR5 and PD-1 and antigen-specific IL-21 production. Tfh cells appear to develop in association with increased Bcl-6 transcription, which suppresses canonical T helper cell developmental pathways (that is, Th1, Th2, Th17, etc.). Though peripheral Tfh cells are not well defined, we have observed that there are subsets of circulating T-cells with a central memory phenotype that express high levels of CXCR5, SLAM (CD150), and CCR6, and dim or low expression of PD-1. These are associated with the magnitude and breadth of class-switch recombination in T-B cell co-culture experiments, decrease with HIV disease progression, and may be lost early during the course of HIV infection [19, 20].
The cellular immunology of hepatitis vaccine response in treated and/or well-controlled HIV infection is not well studied and may provide further insight into the optimization of antigen dose, adjuvant selection, and dosing interval. Here we examined T- and B-cell phenotype and function and plasma cytokine profiles in a well-defined cohort of HIV-infected hepatitis B vaccine responders and non-responders [4] in order to characterize the nature and quality of the immune response to HIV-infection and HBV, with the goal of identifying some of the basic mechanisms of B-cell and T-cell biology. We hypothesized that preserved cellular immune phenotype profile and functionality (as measured by antigen-specific cytokine production) are associated with HBV response.
Materials and Methods
Study Subjects
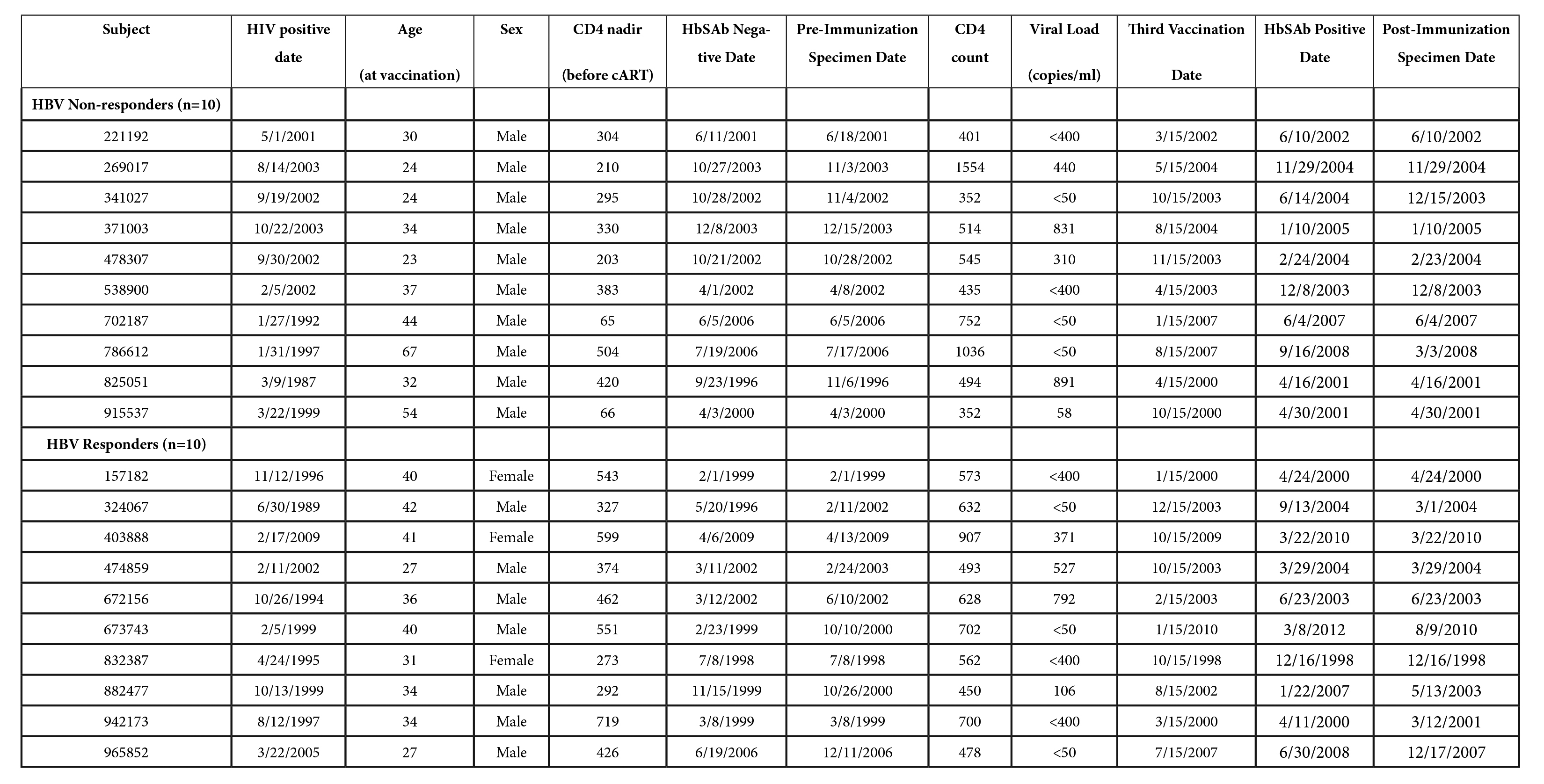
Twenty individuals were identified from the U.S. Military HIV Natural History Study cohort [21, 22] with the following inclusion criteria: (1) past or current use of cART; (2) CD4 count (> 350 cells/µl); (3) no evidence of hepatitis B infection (hepatitis B surface antigen, surface antibody, and core antibody negative); (4) with concurrent frozen peripheral blood mononuclear cells (PBMC) and plasma collected prior to hepatitis B immunization (standard immunization schedule) and within 12 months after the third vaccination. All subjects provided informed consent prior to participation in the prospective cohort study for which collection of stored blood specimens was utilized for performance of assays in this study. Human experimentation guidelines of the United States Department of Health and Human Services were followed in the conduct of clinical research, under a protocol reviewed and approved by the Institutional Review Boards of the National Institutes of Health and Uniformed Services University, both in Bethesda, Maryland.
Laboratory Assays
Polychromatic flow cytometry.
Phenotypic analysis:
PBMCs were cultured in RPMI 1640 (Invitrogen) supplemented with 10%
fetal bovine serum, 2mM L-glutamine, 100U/mL penicillin, and 100 ug /mL
streptomycin (Invitrogen). 1-2 x 106 PBMCs were incubated
with Aqua viability dye and surface stained with titrated amounts of
antibodies to panel (1): CD3; CD4; CD27; CD45RA; CD127; CCR7; PD-1;
CCR6; CXCR5; and ICOS followed by intracellular staining for CTLA-4 and
Ki-67 or (2) CD3; CD19; CD20; CD27; CD38; IgD; IgG; IgM; CXCR5; and
CCR7. Intracellular cytokine staining: 3 x 106
PBMCs were rested for two hours, then incubated for six hours in 1mL of
medium containing brefeldin A (10ug/mL) in the absence or presence of
HIV-1 subtype B Gag-peptide pools (15mers overlapping by 11 residues;
National Institutes of Health AIDS Research and Reference Reagent
Program), hepatitis B surface antigen peptide pools (hepatitis B virus
Genotype A2/strain adw2, 15 mers overlapping by 11 residues, Proimmune,
Sarasota, FL, USA), or Cytostim™ 20µl/ml PBMC suspension (positive
control for TCR activation and intracellular cytokine production)
(Miltenyi Biotech, Auburn, CA). After washing, cells were surface
stained with Aqua, CD4, CD8, CD27, CD45RO, CD127, PD-1, and CXCR5.
Cells were then washed again, permeabilized (Cytofix/Cytoperm kit; BD
Biosciences), and stained with antibodies to CD3, CD154, IFN-γ, IL-2,
TNF-
Antibodies.
Flow cytometry was performed using the following directly conjugated
antibodies: (1) BD Biosciences: CD3 H7APC (SK7), CD45RA Cy7PE (L48),
CD20 APC-H7 (2H7), CD45RO TRPE (UCHL), CTLA-4 APC (BNI3), IgD PE
(IA6-2), IgG APC (G18-145), IgM PE-Cy5 (G20-127), IL-21 Alexa 647
(3A3-N2.1), and Ki-67 Alexa700 (150503); (2) Beckman Coulter: CD127
Cy5PE (R34.34); (3) BioLegend: PD-1 BV711 (EH12.2H7), IFNg BV421
(4S.B3), TNF-
Measurement of plasma cytokines and antibody titers.
Plasma or serum samples from subjects at the same time points before
and after vaccination were measured for soluble CD14 (sCD14), CXCL-13
using standard ELISA assays according to manufacturer’s instructions
(Quantikine® ELISA, R & D Systems, Minneapolis, MN) and
for the following chemokines and cytokines: IL-2, IL-4, IL-6, IL-10,
IL-12p70, IL-15, IL-17A, IL-21, TNF-
Statistical analysis. Experimental variables were analyzed using nonparametric statistical tests of inference: Mann-Whitney U test; the Wilcoxon matched-pairs signed rank test; or the Kruskal-Wallis test with Dunn’s multiple comparison post-test as appropriate. Correlation analysis was performed using the nonparametric Spearman test. Statistical analyses were performed with GraphPad Prism (GraphPad Software, version 5.0) or Stata Statistical Software, Release 11 (StataCorp, College Station, TX).
Results
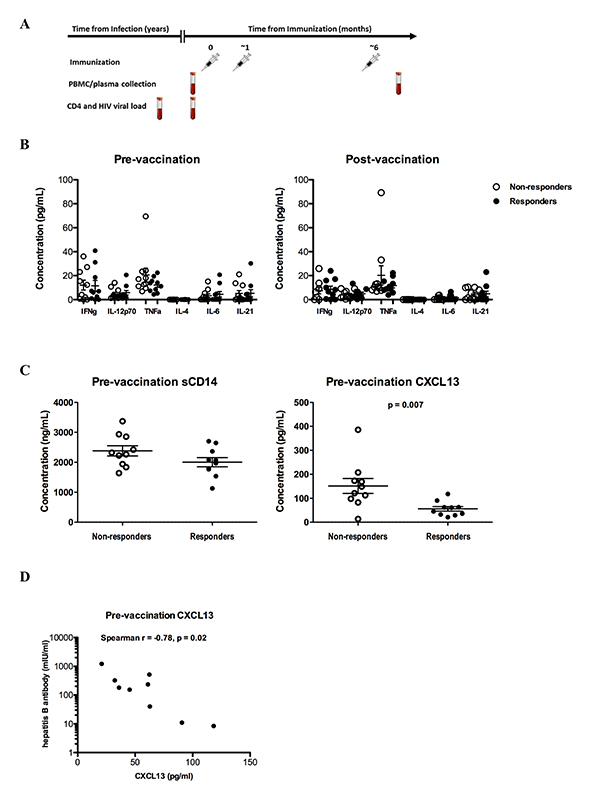
Baseline plasma levels of CXCL-13 are a correlate of vaccine-elicited antibody titers
We utilized a natural history cohort of HIV-infected participants in a
longitudinal study that has prospectively enrolled ~5,000 subjects
since 1986 to identify individuals on treatment, with appropriately
timed blood draws corresponding to hepatitis B vaccination that
included both plasma and PBMC specimens [21, 23, 24]. HIV-infected subjects included in this analysis (Figure 1A and
Supplementary Table 1), classified as hepatitis B vaccine (HBV)
responders (n = 10) and non-responders (n = 10), were not statistically
different in terms of, age, median CD4 count (median 600.5 vs 504), HIV
viral RNA level (median log10 copies/mL, 1.69 vs 1.73), or duration of
HIV infection (mean 40.0 months vs 43.1), but were mostly men (all
non-responders and 7 responders, P = 0.2, Fisher’s exact test).
A similar profile of plasma cytokines was found between the
non-responders and responders both pre- and post-vaccination (Figure 1B
and Supplementary Figure 1). Although not significantly, increased
levels of the pro-inflammatory cytokine tumor necrosis factor-alpha
(TNF-
Figure 1. Baseline plasma CXCL-13 levels predict the response to HBV vaccination (A) HIV-infected subjects with and without detectable antibody responses were assessed before and after hepatitis B vaccination, (B) Pooled data showing the levels of several cytokines measured in plasma samples from non-responders (n = 10) and responders (n = 10), (C) soluble CD14 levels in non-responders and responders are shown in the left panel, while circulating levels of the chemokine CXCL-13 pre-vaccination are shown in the right panel, (D) hepatitis B surface antibody (HbSAb) titer among responders post-vaccination. Figures are scatterplots with mean and SEM with only statistically significant P-values shown as determined by the Mann-Whitney U test (C), and Spearman rank correlation (D).
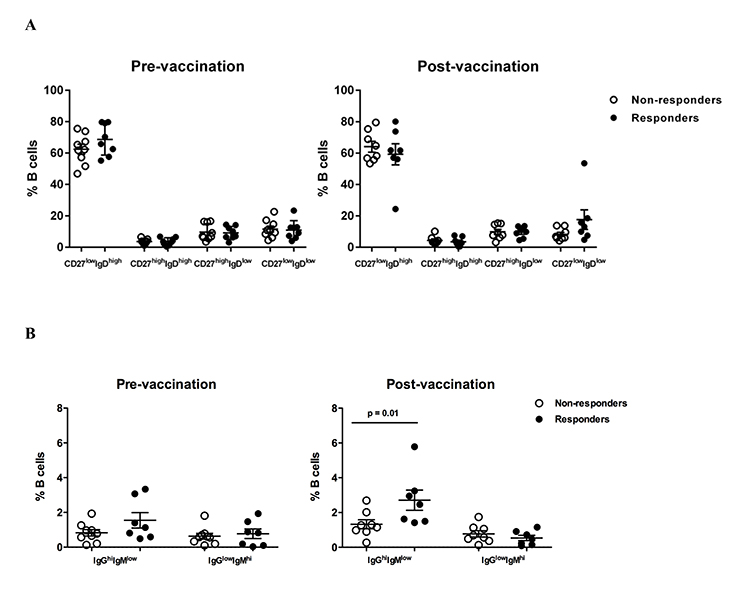
Vaccine response is associated with increased IgGhigh memory B cells post vaccination
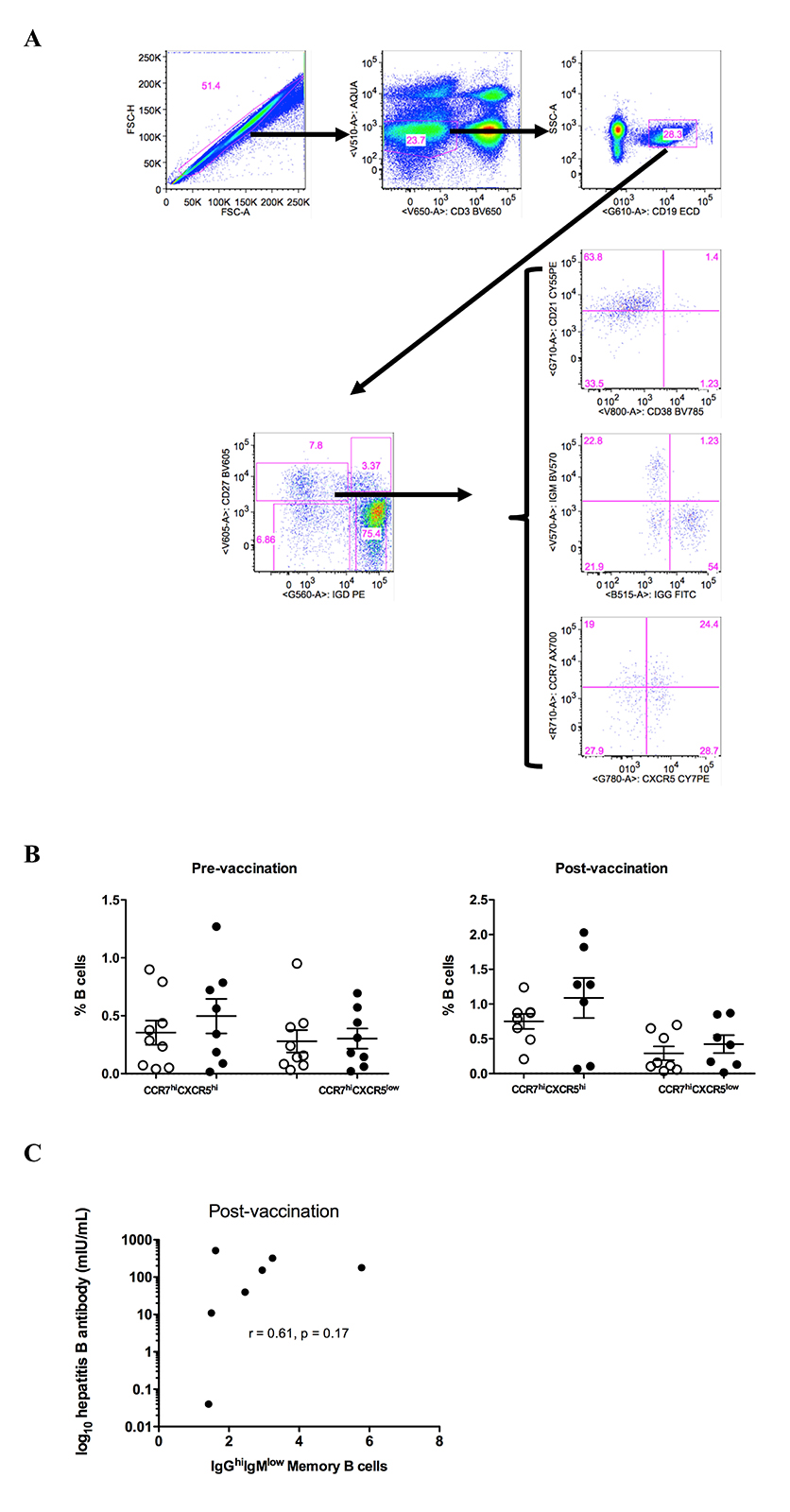
Next, the relative frequencies of circulating B cell populations were investigated pre- and post-vaccination. No differences in terms of B cell differentiation, judged by CD27 and IgD expression, were found between non-responders or responders both pre- and post-vaccination (Figure 2A and Supplementary Figure 2A). No differences were found between responders and non-responders when B-cell populations defined by the expression of chemokine receptors (CCR7, CXCR5) important for B-cell trafficking in LNs [27, 28] (Supplementary Figure 2A and B) or CD21 (or CD27) and CD38 were analyzed (data not shown). Although no difference was found pre-vaccination, a significantly higher (P = 0.01) frequency of circulating IgGhighIgMlowCD27highIgD low “switched” memory B cells was found in responders compared to non-responders post-vaccination (Figure 2B, gating shown in Supplementary Figure 2A). Although the cohort size was underpowered to assess the potential correlation, a positive association between IgG highIgMlowCD27highIgDlow B cells and vaccine titer was found (Supplementary Figure 2C). Our data indicate that vaccine responses are associated with induction of particular memory B cells capable of producing antibodies.
Figure 2. HBV response is associated with increased frequency of IgGhigh memory B cells post vaccination. (A) Pooled data showing the relative frequencies of B-cell populations, judged by the expression of CD27 and IgD, in non-responders and responders pre- and post-vaccination, (B) the relative frequencies of IgGhighIgMlow switched memory (CD27highIgDlow) B cells pre- and post-vaccination. Data presented in Figures A and B are scatterplots with mean and SEM with P-values calculated using Mann-Whitney U test.
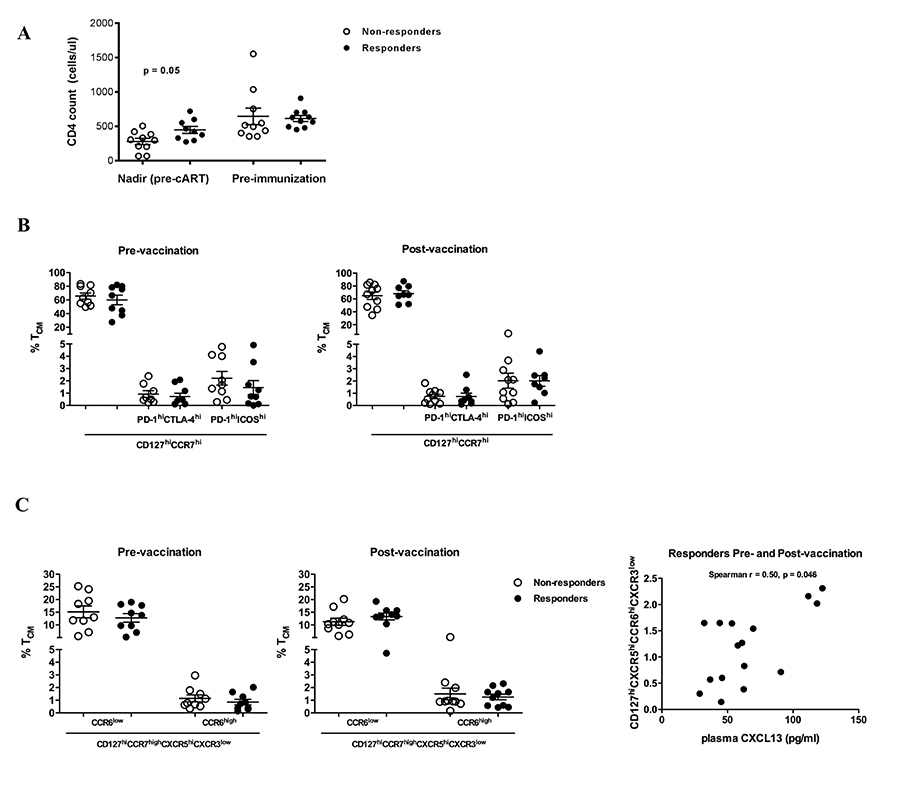
Circulating Tfh cells correlate with plasma levels of CXCL-13 in HBV responders
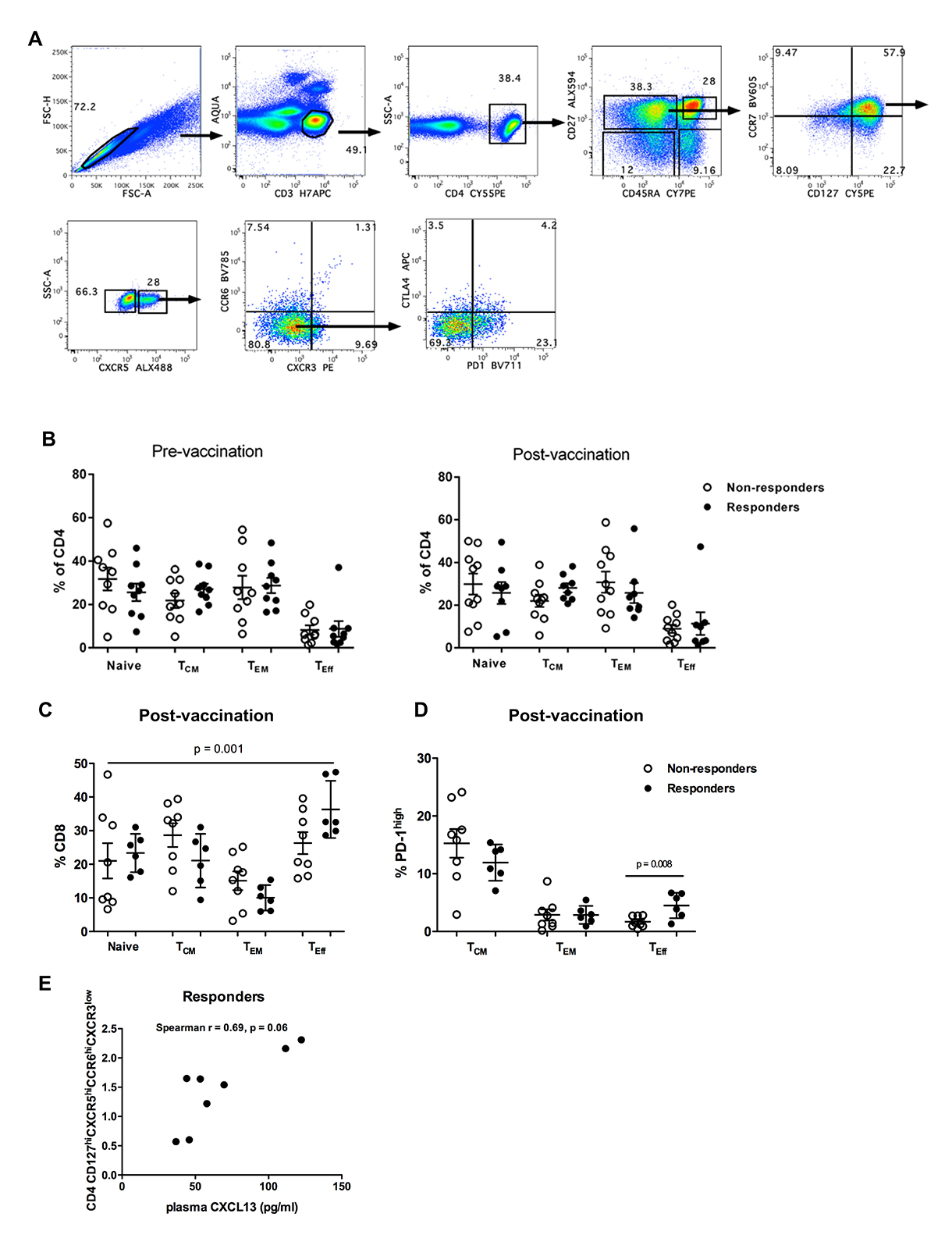
Next the relative frequencies of circulating CD4 T cell subsets with potential B-cell help function [19] was analyzed. Although absolute CD4 counts didn’t differ between non-responders and responders pre-vaccination, a significantly lower nadir CD4 count (pre-cART) was found in non-responders compared to responders (Figure 3A). The frequencies of memory populations defined by flow cytometry staining for CD27 and CD45RA (Supplementary Figure 3A) were similar between responders and non-responders pre- and post-vaccination for CD4 (Supplementary Figure 3B). However, post-vaccination CD8 memory T cells showed a skewed phenotype (a more differentiated, maturation profile of “memory effector-TEff ”) in responders (P = 0.001, Kruskal-Wallis test) (Supplementary Figure 3C). CD4 central memory population subsets (T CM, CD27highCD45RAlow), to include phenotypes with an “activation” profile (PD-1highICOS high or PD-1highCTLA-4high), were not associated with vaccine response (Figure 3B). Next, the profiles of circulating CD4 T cell follicular helper-like cells (CD127highCCR7highCXCR5highCCR6 high/lowCXCR3low, pTfh) [19, 29] were analyzed. Contrary to responders, a trend for reduced frequency of CCR6low pTfh cells after vaccination was observed in non-responders (P = 0.07, Wilcoxon matched pairs test) (Figure 3C, left panel). Although similar frequencies of CCR6 high pTfh cells were found between non-responders and responders (Figure 3C, left panel) a significant correlation was found between plasma CXCL-13 levels and the frequency of CCR6high pTfh cells only in responders (Spearman r = 0.50, P = 0.046) (Figure 3C, right panel and Supplementary Figure 3E).
Figure 3. Circulating pTfh subpopulations correlate with plasma levels of CXCL-13 in HBV vaccine responders. (A) Absolute counts of CD4 T cells in non-responders and responders pre-vaccination are shown, (B) pooled data showing the relative frequencies of “activated” central memory CD127 highCCR7high CD4 T-cell subpopulations in non-responders and responders pre- and post-vaccination, (C) Pooled data showing the relative frequencies of central memory CD27high CD45RAhighCD127highCCR7high CD4 T-cell populations with respect to expression of CCR6 (left panel) and the correlation between CCR6high pTfh cells and plasma levels of CXCL-13 is shown for vaccine responders (right panel). Scatterplots with mean and SEM denoted with statistical analysis performed using Mann-Whitney U test. Correlations with P-values determined using Spearman’s rank correlation.
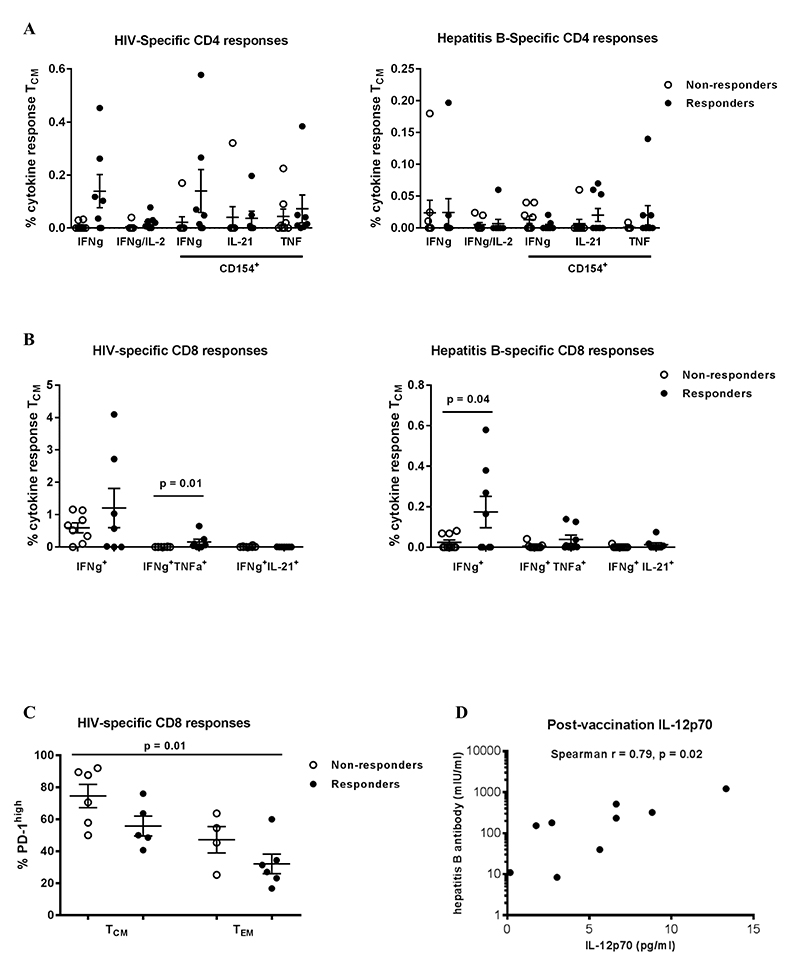
Preserved Th1 adaptive responses in HBV vaccine responders
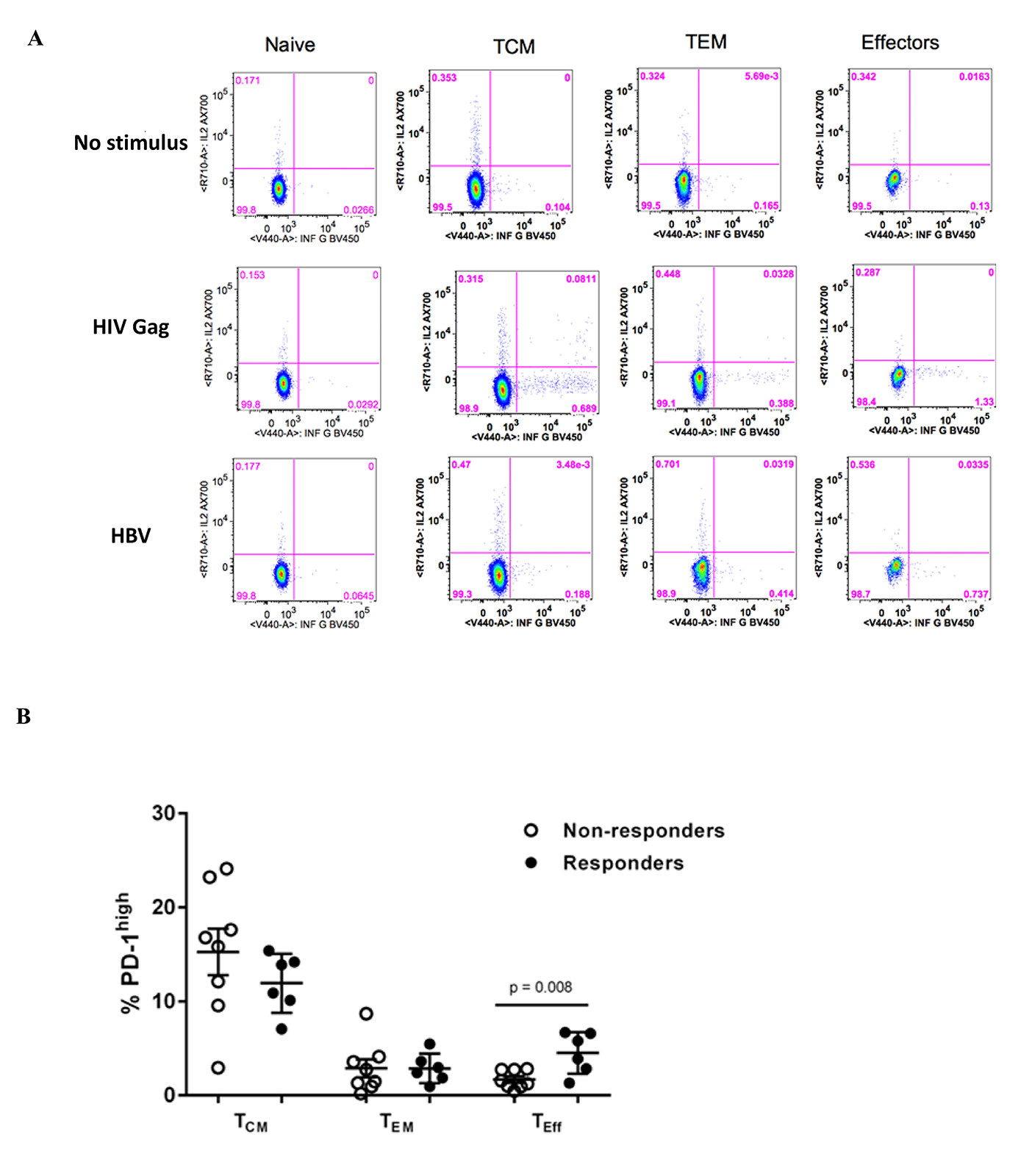
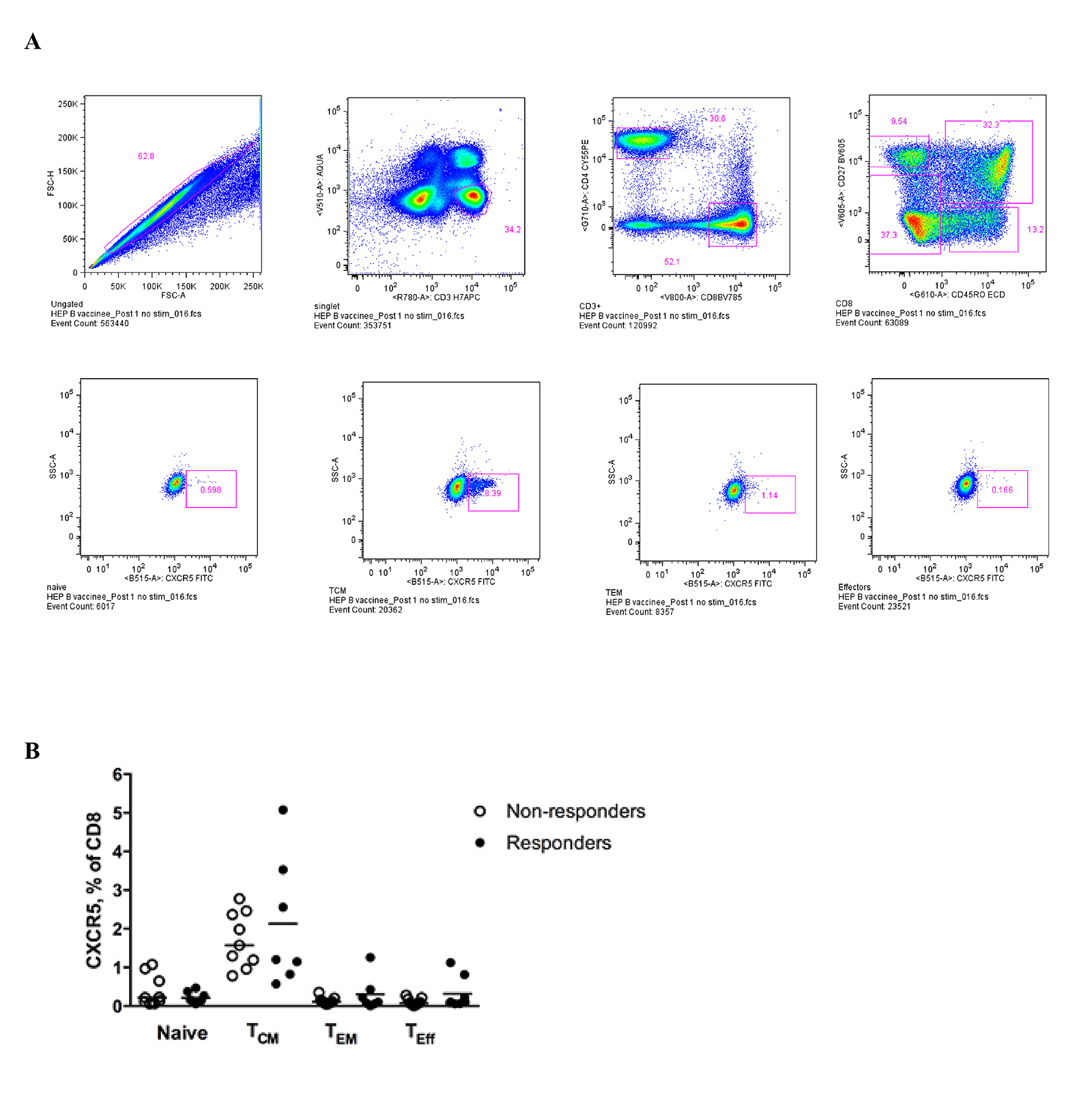
We assessed T-cell function by measuring cytokine production after short (6 hours) ex vivo stimulation with HIV Gag and hepatitis B surface antigen pooled peptides (Supplementary Figure 4A). HIV-specific, but not hepatitis B-specific, CD4 T cells were observed more frequently after stimulation with HIV Gag or hepatitis B surface antigen peptides (Figure 4A). Although not statistically significant (P = 0.09), we observed an increased frequency of HIV-specific CD4 T cells among vaccine responders compared to non-responders (Figure 4A). These responses were predominantly CD27highCD45RO high (data not shown). Both hepatitis B- and HIV-specific CD8 T cell responses were observed more frequently among HBV responders (Figure 4B). Virus-specific responses were observed more frequently in the T central memory compartment (TCM, CD27high CD45ROhigh) compared to more differentiated T cells (CD27 low, data not shown). However, the frequencies of these virus-specific responses were not associated with the magnitude of the vaccine response (data not shown). PD-1 expression was higher on bulk, less differentiated memory CD8 T cells (TCM cells, CD27highCD45RAlow) while significantly higher PD-1 expression was observed on “effector memory” CD8 T cells (CD27lowCD45RAlow) among vaccine responders (P = 0.008, Mann-Whitney test) (Supplementary Figure 4B). A trend for higher PD-1 expression was observed for HIV-specific responses among vaccine non-responders, but was not statistically significant for comparison within populations (eg, TCM for responders compared to non-responders), but was statistically significant across populations (P = 0.01, Kruskal-Wallis test) (Figure 4C). Although plasma IL-12p70, a major Th1 cytokine [30], was not associated with vaccine response, it was associated with the titer magnitude when measured post-vaccination (Spearman r = 0.79, P = 0.02) (Figure 4D). In light of recent observations of follicular CD8 T cell dynamics in chronic viral infection [31-33], we examined CXCR5 expression on CD8 T cell memory subsets and observed increased expression on TCM compared to other subsets, but there was no difference between responders and non-responders (Supplementary Figure 5A and B). Our data point to a better-preserved adaptive antigen-specific Th1 cell immunity in vaccine responders compared to non-responders.
Figure 4. Preserved Th1 adaptive immunity in HBV vaccine responders. (A) Pooled data showing the relative frequencies of antigen-specific CD4 T cell responses measured by intracellular cytokine staining (ICS) after short ex vivo stimulation with HIV Gag (left panel) or hepatitis B surface antigen peptides (right panel), (B) Pooled data showing the relative frequencies of antigen-specific CD8 T cell responses measured with ICS, (C) expression of PD-1 in HIV-specific CD8 T cells in non-responders and responders (P = 0.001, Kruskal-Wallis test), (D) Correlation between plasma IL-12p70 levels and HbSAb titer. Data presented are scatterplots with mean and SEM with P-values calculated using the Mann-Whitney U test (B) or Spearman correlation test (D).
Discussion
We set out to explore reasons why HIV-infected patients, with preserved or reconstituted CD4 counts > 350 cells/µl on treatment with suppressed or low viral load fail to respond adequately to standard dose and schedule hepatitis B vaccine (three doses on a 0-, 1-, and 6-month schedule of either 20 µg Engerix-B® or 10 µg Recombivax-HB®) or maintain durable responses [5, 34, 35]. This phenomenon is observed frequently in the clinic, common in HIV infection and hemodialysis patients, and has resulted in clinical trials of alternative immunization regimens or with higher antigen dose and number of doses administered [11, 36, 37]. We observed that an immune profile prior to and/or after vaccination that is consistent with better Th1 responses was found more commonly in hepatitis B vaccine responders.
Similar general immune activation judged by sCD14 levels was found between non-responders and responders. Despite the similar immune activation, significantly higher levels of CXCL-13, a biomarker of germinal center reactions [26], were found in vaccine non-responders before vaccination, indicating a selective hyperactivity of germinal center immune reactivity in the non-responders. The similar levels of several circulating cytokines tested further support this selective profile. Furthermore, the levels of CXCL-13 were a predictor of the vaccine-induced antibody titers. Therefore, pre-vaccination vigorous germinal center reactions may negatively impact the development of HBV vaccine-induced specific humoral responses. Investigation of the relative impact of residual actively transcribed virus in lymph node areas compared to local inflammatory signals on this profile could lead to novel ways for improvement of HBV vaccine efficiency in cART-treated individuals. Whether this profile is specific for HBV vaccination or applies to other immunogens/antigens is not known and needs further investigation.
Interestingly, HBV vaccine significantly induced the circulating memory B cells expressing an IgG+ phenotype positively associated with the vaccine-induced antibody titers. Among all B-cell populations tested, this was the only population significantly affected, indicating that the HBV vaccine induced specific B cell populations in responders. No correlation between circulating CXCL-13 and vaccine-induced titers post-vaccination was found, possibly due to the time point plasmas were collected post-vaccination (7 days post-vaccination would be an appropriate time point for this purpose). However, a significant correlation was found between CXCL-13 and circulating CD4 T cell populations with increased capacity for B-cell help [19, 29] in vaccine-responders, further supporting the use of plasma CXCL-13 levels as a biomarker for germinal reactions [26]. Therefore, HBV vaccine changes the dynamics of relevant populations related to GC reactions in the responders group. Further studies, utilizing larger cohorts of relevant individuals, will establish whether such populations can be used as “biomarkers” for HBV vaccine responsiveness.
An overall better preserved antigen-specific adaptive Th1 cell immunity was found in responders compared to non-responders, especially in the antigen-specific CD8 T cell compartment for both HIV and vaccine antigens. This profile was also associated with fewer bulk and virus-specific CD8 T cells expressing an “exhausted” phenotype. The association between better preserved Th1 responses and vaccine responsiveness was further supported by the significant correlation between HBV vaccine–induced antibody titers and circulating levels of IL-12p70, a prototype member of Th1 cytokines [30]. These differences in cellular immune response may reflect earlier treatment, a higher CD4 nadir prior to treatment, or shorter duration of infection, though the two groups compared for these analyses were similar in this regard (Supplementary Table 1) [38-40]. More recently, follicular CXCR5high CD8 T cells have been implicated in the control of chronic viral infection in lymphatic tissue [31, 33], to include HIV infection [32]. Differences in CXCR5 expression on peripheral T cells have been observed in non-infectious causes of altered T cell dynamics (autoimmunity and primary immunodeficiency) [41, 42]. Furthermore, recent studies have shown an increased frequency of CXCR5high CD8 T cells in the lymph node follicular areas in chronic HIV [32] and SIV [43] infection. We evaluated CXCR5 expression on peripheral CD8 T cells and observed similarly increased expression in the “central memory” CD8 T cells in responders and non-responders. Further investigation is needed to confirm whether i) the better preserved circulating virus-specific CD8 T cell responses found among responders also reflect more “efficient” HIV-specific CD8 T cell immunity in the lymph nodes and follicular areas, a “sanctuary area” for HIV [31, 33], and ii) the frequency of circulating CXCR5high CD8 T cells could be used as a surrogate for the accumulation of CXCR5 high CD8 T cells in the follicles.
In this study we did not investigate the duration of the antibody response, although one of our HBV responders had an HbSAb concentration of less than 10 mIU/mL approximately a year after completion of vaccination. This is an area of further investigation, although it is unclear whether maintenance of an adequate HbSAb concentration, once achieved, is associated with breakthrough HIV infection in treated individuals [5, 35]. Furthermore, the small sample size, given the characteristics of the subjects identified for study and the availability of archived specimens for testing, did not allow for further analysis of T-cell populations that could have an impact on the development of B-cell responses. However, our study points to possible mechanisms for the responsiveness to HBV vaccination in HIV infected individuals [4, 5] even after cART treatment [4, 6] and provides a rational base for future studies focusing on the germinal center dynamics that could direct HBV vaccine efficacy.
Potential Conflicts of Interest
The authors declare no competing financial interests.
Financial Support
This research was supported by the Intramural Research Program of the Vaccine Research Center, National Institutes of Allergy and Infectious Diseases, and the National Institutes of Health. Support for this work (IDCRP-000-02) was also provided by the Infectious Disease Clinical Research Program (IDCRP), a Department of Defense (DoD) program executed through the Uniformed Services University of the Health Sciences. This project has been funded in whole, or in part, with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health (NIH), under Inter-Agency Agreement Y1-AI-5072. LGM received a scholarship support from Conselho Nacional de Desenvolvimento Cientifico e Tecnológico (CNPq-PDE, process # 0722778224537798), Brazil.
Disclaimer
The opinions herein are those of the authors and should not be construed as official or representing the views of the U.S. Department of Health and Human Services, the National Institute of Allergy and Infectious Diseases, the Department of Defense, the Department of the Army, the Uniformed Services University of the Health Sciences, or the Henry M. Jackson Foundation for the Advancement of Military Medicine.
References
1. Poland GA, Jacobson RM. Clinical practice: prevention of hepatitis B with the hepatitis B vaccine. N Engl J Med. 2004;351(27):2832-8. PubMed PMID: 15625334. doi: 10.1056/NEJMcp041507
2. Alper CA, Kruskall MS, Marcus-Bagley D, Craven DE, Katz AJ, Brink SJ, Dienstag JL, Awdeh Z, Yunis EJ. Genetic prediction of nonresponse to hepatitis B vaccine. N Engl J Med. 1989;321(11):708-12.
3. Hohler T, Reuss E, Evers N, Dietrich E, Rittner C, Freitag CM, Vollmar J, Schneider PM, Fimmers R. Differential genetic determination of immune responsiveness to hepatitis B surface antigen and to hepatitis A virus: a vaccination study in twins. Lancet. 2002;360(9338):991-5.
4. Landrum ML, Hullsiek KH, Chun HM, Crum-Cianflone NF, Ganesan A, Weintrob AC, Barthel RV, O’Connell RJ, Agan BK. The timing of hepatitis B virus (HBV) immunization relative to human immunodeficiency virus (HIV) diagnosis and the risk of HBV infection following HIV diagnosis. Am J Epidemiol. 2011;173(1):84-93. PubMed PMID: 21051446. Pubmed Central PMCID: 3025642. doi: 10.1093/aje/kwq326
5. Landrum ML, Hullsiek KH, Ganesan A, Weintrob AC, Crum-Cianflone NF, Barthel RV, O’Connell RJ, Fieberg A, Chun HM, Marconi VC, Dolan MJ, Agan BK. Hepatitis B vaccination and risk of hepatitis B infection in HIV-infected individuals. AIDS. 2010;24(4):545-55. PubMed PMID: 19487908. Pubmed Central PMCID: 2831117. doi: 10.1097/QAD.0b013e32832cd99e
6. del Pozo Balado Mdel M, Leal M, Mendez Lagares G, Mata RC, Lopez-Cortes LF, Viciana P, Pacheco YM. Increased regulatory T cell counts in HIV-infected nonresponders to hepatitis B virus vaccine. J Infect Dis. 2010;202(3):362-9. PubMed PMID: 20560766. doi: 10.1086/653707
7. Kroon FP, van Dissel JT, de Jong JC, Zwinderman K, van Furth R. Antibody response after influenza vaccination in HIV-infected individuals: a consecutive 3-year study. Vaccine. 2000;18(26):3040-9. PubMed PMID: 10825608.
8. Launay O, Desaint C, Durier C, Loulergue P, Duval X, Jacomet C, Pialoux G, Ghosn J, Raffi F, Rey D, Ajana F, Colin de Verdière N, Reynes J, Foubert V, Roman F, Devaster JM, Delfraissy JF, Aboulker JP. Safety and Immunogenicity of a Monovalent 2009 Influenza A/H1N1v Vaccine Adjuvanted With AS03A or Unadjuvanted in HIV-Infected Adults: A Randomized, Controlled Trial. J Infect Dis. 2011;204(1):124-34. PubMed PMID: 21628666. Pubmed Central PMCID: 3307156. doi: 10.1093/infdis/jir211
9. Pallikkuth S, Parmigiani A, Silva SY, George VK, Fischl M, Pahwa R, Pahwa S. Impaired peripheral blood T follicular helper cell function in HIV-infected nonresponders to the 2009 H1N1/09 vaccine. Blood. 2012;120(5):985-93. PubMed PMID: 22692510. doi: 10.1182/blood-2011-12-396648
10. Tasker SA, Treanor JJ, Paxton WB, Wallace MR. Efficacy of influenza vaccination in HIV-infected persons. A randomized, double-blind, placebo-controlled trial. Ann Intern Med. 1999;131(6):430-3. PubMed PMID: 10498559.
11. Launay O, van der Vliet D, Rosenberg AR, Michel ML, Piroth L, Rey D, Colin de Verdi ère N, Slama L, Martin K, Lortholary O, Carrat F, Trial AHV-B. Safety and immunogenicity of 4 intramuscular double doses and 4 intradermal low doses vs standard hepatitis B vaccine regimen in adults with HIV-1: a randomized controlled trial. JAMA. 2011;305(14):1432-40. PubMed PMID: 21486976. doi: 10.1001/jama.2011.351
12. Malaspina A, Moir S, Orsega SM, Vasquez J, Miller NJ, Donoghue ET, Kottilil S, Gezmu M, Follmann D, Vodeiko GM, Levandowski RA, Mican JM, Fauci AS. Compromised B cell responses to influenza vaccination in HIV-infected individuals. J Infect Dis. 2005;191(9):1442-50. PubMed PMID: 15809902. doi: 10.1086/429298
13. De Milito A, Morch C, Sonnerborg A, Chiodi F. Loss of memory (CD27) B lymphocytes in HIV-1 infection. AIDS. 2001;15(8):957-64. PubMed PMID: 11399977.
14. De Milito A, Nilsson A, Titanji K, Thorstensson R, Reizenstein E, Narita M, Grutzmeier S, Sonnerborg A, Chiodi F. Mechanisms of hypergammaglobulinemia and impaired antigen-specific humoral immunity in HIV-1 infection. Blood. 2004;103(6):2180-6. PubMed PMID: 14604962. doi: 10.1182/blood-2003-07-2375
15. Cagigi A, Nilsson A, Pensieroso S, Chiodi F. Dysfunctional B-cell responses during HIV-1 infection: implication for influenza vaccination and highly active antiretroviral therapy. Lancet Infect Dis. 2010;10(7):499-503. PubMed PMID: 20610332. doi: 10.1016/S1473-3099(10)70117-1
16. Moir S, Ho J, Malaspina A, Wang W, DiPoto AC, O’Shea MA, Roby G, Kottilil S, Arthos J, Proschan MA, Chun TW, Fauci AS. Evidence for HIV-associated B cell exhaustion in a dysfunctional memory B cell compartment in HIV-infected viremic individuals. J Exp Med. 2008;205(8):1797-805. PubMed PMID: 18625747. Pubmed Central PMCID: 2525604. doi: 10.1084/jem.20072683
17. He B, Qiao X, Klasse PJ, Chiu A, Chadburn A, Knowles DM, Moore JP, Cerutti A. HIV-1 envelope triggers polyclonal Ig class switch recombination through a CD40-independent mechanism involving BAFF and C-type lectin receptors. J Immunol. 2006;176(7):3931-41. PubMed PMID: 16547227.
18. Vinuesa CG, Cyster JG. How T cells earn the follicular rite of passage. Immunity. 2011;35(5):671-80. PubMed PMID: 22118524. doi: 10.1016/j.immuni.2011.11.001
19. Boswell KL, Paris R, Boritz E, Ambrozak D, Yamamoto T, Darko S, Wloka K, Wheatley A, Narpala S, McDermott A, Roederer M, Haubrich R, Connors M, Ake J, Douek DC, Kim J, Petrovas C, Koup RA. Loss of Circulating CD4 T Cells with B Cell Helper Function during Chronic HIV Infection. PLoS Pathog. 2014;10(1):e1003853. PubMed PMID: 24497824. doi: 10.1371/journal.ppat.1003853
20. Paris RM, Petrovas C, Ferrando-Martinez S, Moysi E, Boswell KL, Archer E, Yamamoto T, Ambrozak D, Casazza JP, Haubrich R, Connors M, Ake J, Kim JH, Koup RA. Selective Loss of Early Differentiated, Highly Functional PD1high CD4 T Cells with HIV Progression. PLoS One. 2015;10(12):e0144767. PubMed PMID: 26678998. doi: 10.1371/journal.pone.0144767
21. Okulicz JF, Marconi VC, Landrum ML, Wegner S, Weintrob A, Ganesan A, Hale B, Crum-Cianflone N, Delmar J, Barthel V, Quinnan G, Agan BK, Dolan MJ. Clinical outcomes of elite controllers, viremic controllers, and long-term nonprogressors in the US Department of Defense HIV natural history study. J Infect Dis. 2009;200(11):1714-23. PubMed PMID: 19852669. doi: 10.1086/646609
22. Chun HM, Fieberg AM, Hullsiek KH, Lifson AR, Crum-Cianflone NF, Weintrob AC, Ganesan A, Barthel RV, Bradley WP, Agan BK, Landrum ML. Epidemiology of Hepatitis B virus infection in a US cohort of HIV-infected individuals during the past 20 years. Clin Infect Dis. 2010;50(3):426-36. PubMed PMID: 20047484. Pubmed Central PMCID: 2805765. doi: 10.1086/649885
23. Okulicz JF, Mesner O, Ganesan A, O’Bryan TA, Deiss RG, Agan BK. Hepatitis B vaccine responsiveness and clinical outcomes in HIV controllers. PLoS One. 2014;9(8):e105591. PubMed PMID: 25144773. Pubmed Central PMCID: Pmc4140789. doi: 10.1371/journal.pone.0105591
24. Reid MJ, Goetz DW, Zajac RA, Houk RW, Boswell RN. The natural history of human immunodeficiency virus infection in screened HIV positive U.S. Air Force personnel: a preliminary report. J Acquir Immune Defic Syndr. 1988;1(5):508-15.
25. Shive CL, Jiang W, Anthony DD, Lederman MM. Soluble CD14 is a nonspecific marker of monocyte activation. AIDS. 2015;29(10):1263-5. PubMed PMID: 26035325. Pubmed Central PMCID: PMC4452959. doi: 10.1097/QAD.0000000000000735
26. Havenar-Daughton C, Lindqvist M, Heit A, Wu JE, Reiss SM, Kendric K, Belanger S, Kasturi SP, Landais E, Akondy RS, McGuire HM, Bothwell M, Vagefi PA, Scully E, Investigators IPCP, Tomaras GD, Davis MM, Poignard P, Ahmed R, Walker BD, Pulendran B, McElrath MJ, Kaufmann DE, Crotty S. CXCL13 is a plasma biomarker of germinal center activity. Proc Natl Acad Sci U S A. 2016;113(10):2702-7. PubMed PMID: 26908875. Pubmed Central PMCID: PMC4790995. doi: 10.1073/pnas.1520112113
27. Okada T, Ngo VN, Ekland EH, Förster R, Lipp M, Littman DR, Cyster JG. Chemokine Requirements for B Cell Entry to Lymph Nodes and Peyer’s Patches. J Exp Med. 2002;196(1):65-75. doi: 10.1084/jem.20020201
28. Gunn MD, Ngo VN, Ansel KM, Ekland EH, Cyster JG, Williams LT. A B-cell-homing chemokine made in lymphoid follicles activates Burkitt’s lymphoma receptor-1. Nature. 1998;391(6669):799-803. PubMed PMID: 9486651. doi: 10.1038/35876
29. Morita R, Schmitt N, Bentebibel SE, Ranganathan R, Bourdery L, Zurawski G, Foucat E, Dullaers M, Oh S, Sabzghabaei N, Lavecchio EM, Punaro M, Pascual V, Banchereau J, Ueno H. Human blood CXCR5(+)CD4(+) T cells are counterparts of T follicular cells and contain specific subsets that differentially support antibody secretion. Immunity. 2011;34(1):108-21. PubMed PMID: 21215658. Pubmed Central PMCID: 3046815. S1074-7613(10)00491-7 [pii] doi: 10.1016/j.immuni.2010.12.012
30. Hsieh C, Macatonia S, Tripp C, Wolf S, O’Garra A, Murphy K. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science. 1993;260(5107):547-9. doi: 10.1126/science.8097338
31. He R, Hou S, Liu C, Zhang A, Bai Q, Han M, Yang Y, Wei G, Shen T, Yang X, Xu L, Chen X, Hao Y, Wang P, Zhu C, Ou J, Liang H, Ni T, Zhang X, Zhou X, Deng K, Chen Y, Luo Y, Xu J, Qi H, Wu Y, Ye L. Follicular CXCR5-expressing CD8+ T cells curtail chronic viral infection. Nature. 2016. PubMed PMID: 27501245. doi: 10.1038/nature19317
32. Petrovas C, Ferrando-Martinez S, Gerner MY, Casazza JP, Pegu A, Deleage C, Cooper A, Hataye J, Andrews S, Ambrozak D, Del Río Estrada PM, Boritz E, Paris R, Moysi E, Boswell KL, Ruiz-Mateos E, Vagios I, Leal M, Ablanedo-Terrazas Y, Rivero A, Gonzalez-Hernandez LA, McDermott AB, Moir S, Reyes-Terán G, Docobo F, Pantaleo G, Douek DC, Betts MR, Estes JD, Germain RN, Mascola JR, Koup RA. Follicular CD8 T cells accumulate in HIV infection and can kill infected cells in vitro via bispecific antibodies. Sci Transl Med. 2017;9(373). PubMed PMID: 28100833. doi: 10.1126/scitranslmed.aag2285
33. Leong YA, Chen Y, Ong HS, Wu D, Man K, Deleage C, Minnich M, Meckiff BJ, Wei Y, Hou Z, Zotos D, Fenix KA, Atnerkar A, Preston S, Chipman JG, Beilman GJ, Allison CC, Sun L, Wang P, Xu J, Toe JG, Lu HK, Tao Y, Palendira U, Dent AL, Landay AL, Pellegrini M, Comerford I, McColl SR, Schacker TW, Long HM, Estes JD, Busslinger M, Belz GT, Lewin SR, Kallies A, Yu D. CXCR5+ follicular cytotoxic T cells control viral infection in B cell follicles. Nat Immunol. 2016; advance online publication. doi: 10.1038/ni.3543. http://www.nature.com/ni/journal/vaop/ncurrent/abs/ni.3543.html#supplementary-information
34. Mehta N, Cunningham CK, Flynn P, Pepe J, Obaro S, Kapogiannis BG, Bethel J, Luzuriaga K, Adolescent Trials Network for HIVAI. Impaired generation of hepatitis B virus-specific memory B cells in HIV infected individuals following vaccination. Vaccine. 2010;28(21):3672-8. PubMed PMID: 20356567. Pubmed Central PMCID: 2862082. doi: 10.1016/j.vaccine.2010.03.022
35. Launay O, Rosenberg AR, Rey D, Pouget N, Michel ML, Reynes J, Neau D, Raffi F, Piroth L, Carrat F, Group AHV-B. Long-term immune response to hepatitis b virus vaccination regimens in adults with human immunodeficiency virus 1: Secondary analysis of a randomized clinical trial. JAMA Intern Med. 2016;176(5):603-10. PubMed PMID: 27064975. doi: 10.1001/jamainternmed.2016.0741
36. Rey D, Piroth L, Wendling MJ, Miailhes P, Michel ML, Dufour C, Haour G, Sogni P, Rohel A, Ajana F, Billaud E, Molina JM, Launay O, Carrat F. Safety and immunogenicity of double-dose versus standard-dose hepatitis B revaccination in non-responding adults with HIV-1 (ANRS HB04 B-BOOST): a multicentre, open-label, randomised controlled trial. Lancet Infect Dis. 2015;15(11):1283-91. PubMed PMID: 26257021. doi: 10.1016/s1473-3099(15)00220-0
37. Chaiklang K, Wipasa J, Chaiwarith R, Praparattanapan J, Supparatpinyo K. Comparison of immunogenicity and safety of four doses and four double doses vs. standard doses of hepatitis B vaccination in HIV-infected adults: a randomized, controlled trial. PLoS One. 2013;8(11):e80409. PubMed PMID: 24265819. Pubmed Central PMCID: PMC3827227. doi: 10.1371/journal.pone.0080409
38. Migueles SA, Connors M. Success and failure of the cellular immune response against HIV-1. Nat Immunol. 2015;16(6):563-70. PubMed PMID: 25988888. doi: 10.1038/ni.3161
39. Bart PA, Rizzardi GP, Tambussi G, Chave JP, Chapuis AG, Graziosi C, Corpataux JM, Halkic N, Meuwly JY, Munoz M, Meylan P, Spreen W, McDade H, Yerly S, Perrin L, Lazzarin A, Pantaleo G. Immunological and virological responses in HIV-1-infected adults at early stage of established infection treated with highly active antiretroviral therapy. AIDS. 2000;14(13):1887-97. PubMed PMID: 10997391.
40. Angel JB, Parato KG, Kumar A, Kravcik S, Badley AD, Fex C, Ashby D, Sun E, Cameron DW. Progressive human immunodeficiency virus-specific immune recovery with prolonged viral suppression. J Infect Dis. 2001;183(4):546-54. PubMed PMID: 11170979. doi: 10.1086/318547
41. Saito R, Onodera H, Tago H, Suzuki Y, Shimizu M, Matsumura Y, Kondo T, Itoyama Y. Altered expression of chemokine receptor CXCR5 on T cells of myasthenia gravis patients. J Neuroimmunol. 2005;170(1-2):172-8. PubMed PMID: 16214223. doi: 10.1016/j.jneuroim.2005.09.001
42. Payne D, Drinkwater S, Baretto R, Duddridge M, Browning MJ. Expression of chemokine receptors CXCR4, CXCR5 and CCR7 on B and T lymphocytes from patients with primary antibody deficiency. Clin Exp Immunol. 2009;156(2):254-62. PubMed PMID: 19250276. Pubmed Central PMCID: PMC2759473. doi: 10.1111/j.1365-2249.2009.03889.x
43. Mylvaganam GH, Rios D, Abdelaal HM, Iyer S, Tharp G, Mavinger M, Hicks S, Chahroudi A, Ahmed R, Bosinger SE, Williams IR, Skinner PJ, Velu V, Amara RR. Dynamics of SIV-specific CXCR5+ CD8 T cells during chronic SIV infection. Proc Natl Acad Sci U S A. 2017. PubMed PMID: 28159893. doi: 10.1073/pnas.1621418114
Supplementary Materials
Supplementary Table. Characteristics of vaccine cohort subjects
Abbreviations: cART, combination antiretroviral therapy; HBV, hepatitis B vaccine; HbSAb, hepatitis B surface antibodySupplementary Figure 1. Scatterplot comparison of additional cytokines (IL-2, IL-10, and IL-15) for HBV responders compared to non-responders.
Supplementary Figure 2. (A) Gating scheme for phenotypic analysis of B cell subsets, (B) Pooled data showing the relative frequency of circulating memory B cells with respect to expression of CCR7 and CXCR5 chemokine receptors and (C) Scatterplot of HBV antibody concentration and the frequency of IgG highIgMlow switched memory B cells (only 7 HBV responding donors had a measurable response). P-values calculated with Mann-Whitney test and (B) and Spearman correlation test (C).
Supplementary Figure 3. (A) Gating scheme for phenotypic analysis of CD4 and CD8 populations, (B) Pooled data showing the relative frequencies of CD4 memory subsets, judged by the expression of CD27 and CD45RA, (C) Pooled data showing the relative frequencies of CD8 memory subsets, judged by the expression of CD27 and CD45RA, (D) the frequency of PD-1high CD8 T cells in memory compartments post-vaccination is shown, (E) Correlation between CCR6 high pTfh cells and plasma levels of CXCL-13 in responders post-vaccination. Data are comparison scatterplots with mean and SEM. P-values calculated using the Mann-Whitney U test (C) or Spearman rank correlation test (E).
Supplementary Figure 4. (A) Pooled data showing the relative frequencies of pTfh cells with respect to expression of CCR6 and PD-1/ICOS pre- and post-vaccination, (B) Gating scheme for analysis of CD4 and CD8 T-cell function by intracellular cytokine staining using (ICS) after stimulation with pooled HIV Gag or hepatitis B surface antigen peptides, (C) expression of PD-1 in memory CD8 T-cell subsets in non-responders and responders post-vaccination (scatterplot with mean and SEM, Mann-Whitney U test).
Supplementary Figure 5. (A) Gating scheme for phenotypic analysis of CXCR5 on CD8 populations, (B) Pooled data showing the relative frequencies of CXCR5highCD8 T-cell populations in non-responders and responders.